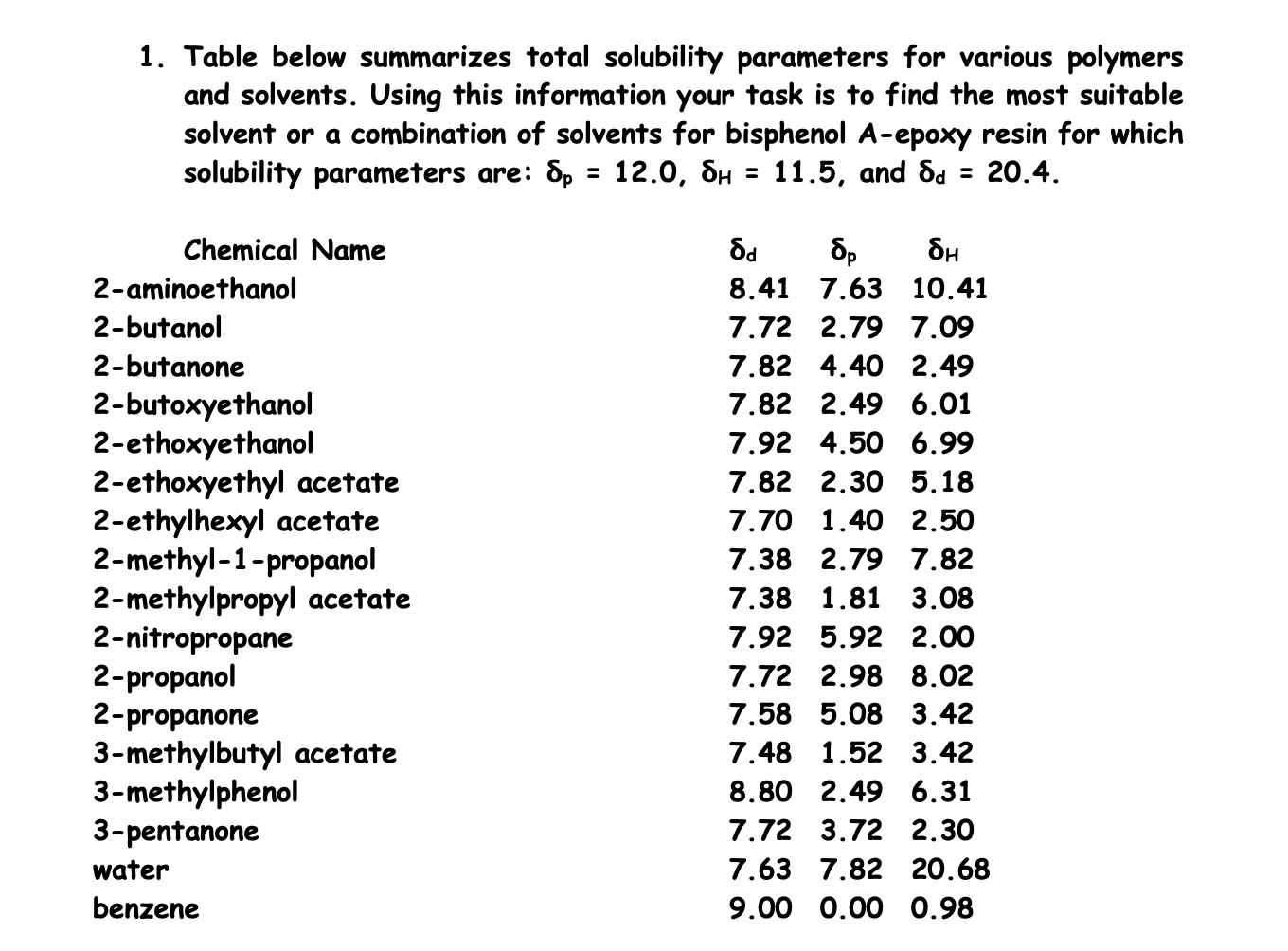
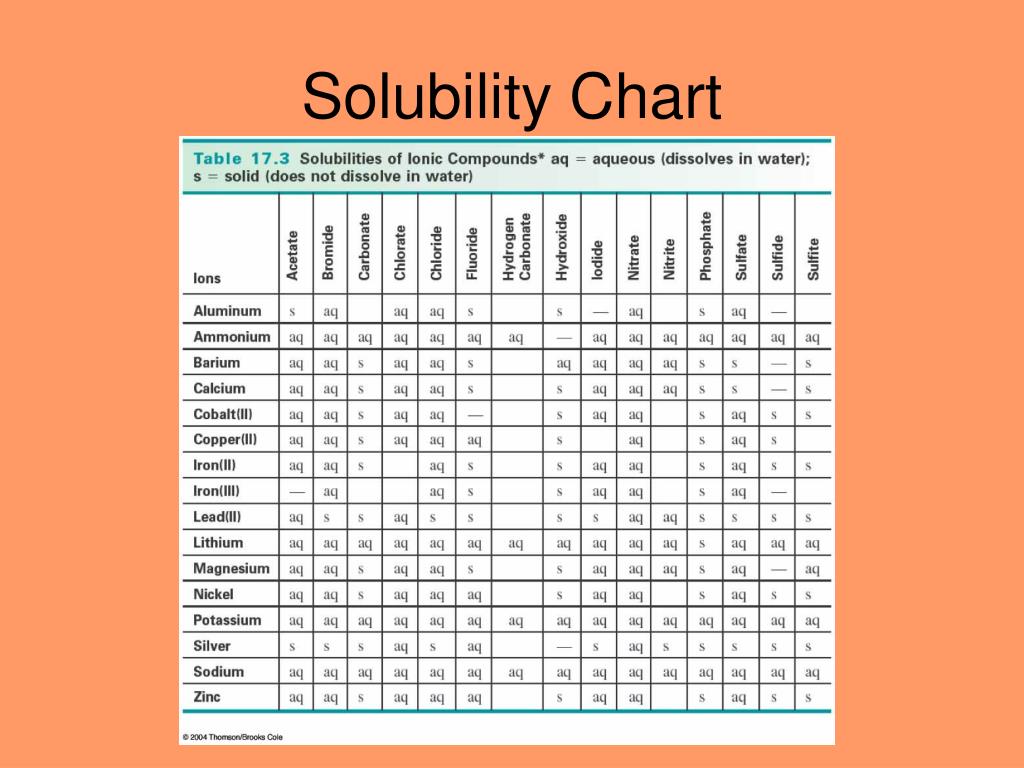
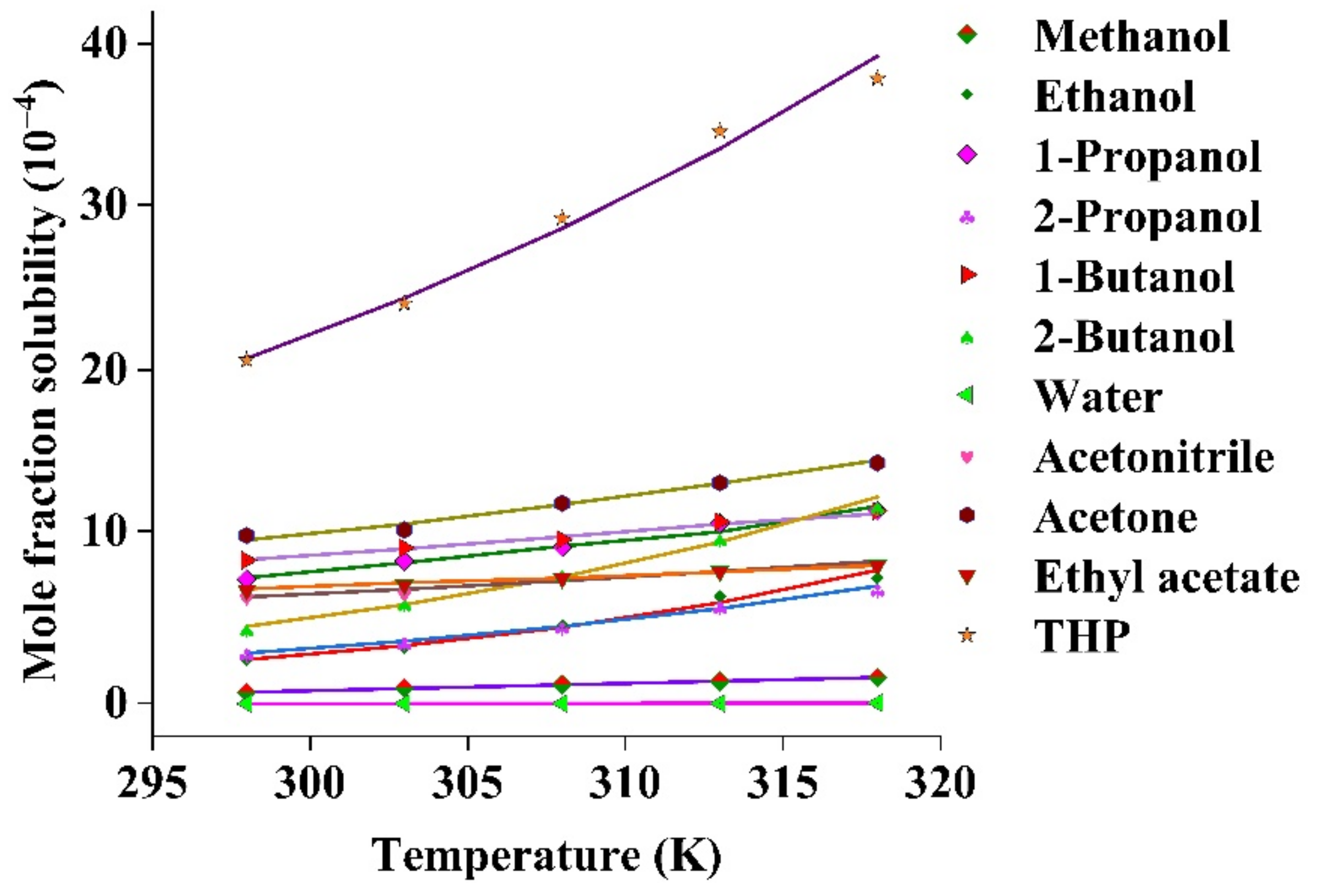
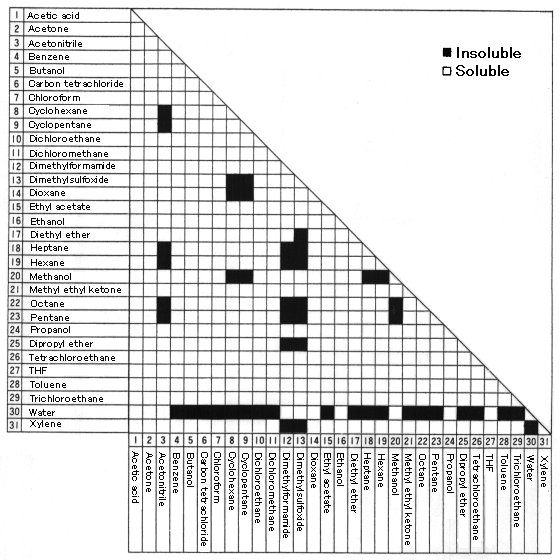
Solvent Solubility Chart
Solvent Solubility Chart - A usually liquid substance capable of dissolving or dispersing one or more other substances. A solvent is a substance that becomes a solution by dissolving a solid, liquid, or gaseous solute. A solvent is usually a liquid, but can also be a solid or gas. A solvent is the component of a solution that is present in the greatest amount. The molecules of the solvent work to put the. A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. It is the substance in which the solute is dissolved. Solvents are often liquids, although they can also be solids, gases, or supercritical fluids. Polar solvents (e.g., water) favour formation of ion s; A solvent is, by definition, any substance that will take other things (aka ‘solutes’) into solution. A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. Solvents are often liquids, although they can also be solids, gases, or supercritical fluids. A solvent is a substance that becomes a solution by dissolving a solid, liquid, or gaseous solute. Water is a solvent for polar molecules, and the most common solvent used by living things; A solvent can be solid, liquid or gas. A solvent is a substance that dissolves a solute to form a homogeneous solution. A solvent is a substance that can dissolve an insoluble solute and create a solution. While most common solvent are liquids, a. Something that provides a solution. A usually liquid substance capable of dissolving or dispersing one or more other substances. Water is a solvent for polar molecules, and the most common solvent used by living things; A solvent can be solid, liquid or gas. For example, in a saltwater mixture, water functions as the solvent, and salt is the solute. A usually liquid substance capable of dissolving or dispersing one or more other substances. A solvent is usually a liquid,. A solvent is a substance that can dissolve an insoluble solute and create a solution. A solvent is a substance that dissolves a solute to form a homogeneous solution. A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. Something that provides a solution. For example, in a saltwater mixture, water functions. A solvent can be solid, liquid or gas. A solvent is usually a liquid, but can also be a solid or gas. A solvent is a substance that can dissolve an insoluble solute and create a solution. For example, in a saltwater mixture, water functions as the solvent, and salt is the solute. Water is a solvent for polar molecules,. A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. A solvent is a molecule that has the ability to dissolve other molecules, known as solutes. A solvent is the component of a solution that is present in the greatest amount. Water is a solvent for polar molecules, and the most common. Essential traits include its dissolving power, homogeneity, and its role as a reaction medium. Something that eliminates or attenuates something. A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. Water is a solvent for polar molecules, and the most common solvent used by living things; It is the substance in which. Solvent, substance, ordinarily a liquid, in which other materials dissolve to form a solution. For example, in a saltwater mixture, water functions as the solvent, and salt is the solute. A solvent is the component of a solution that is present in the greatest amount. A solvent is a substance that dissolves a solute to form a homogeneous solution. Essential. It is the substance in which the solute is dissolved. While most common solvent are liquids, a. Something that eliminates or attenuates something. A usually liquid substance capable of dissolving or dispersing one or more other substances. A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. Something that eliminates or attenuates something. Polar solvents (e.g., water) favour formation of ion s; A solvent is a substance that dissolves a solute to form a homogeneous solution. A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. Usually, a solvent is a liquid. A solvent can be solid, liquid or gas. A solvent is usually a liquid, but can also be a solid or gas. A solvent is the component of a solution that is present in the greatest amount. It is the substance in which the solute is dissolved. A solvent is a substance that becomes a solution by dissolving a solid,. Something that provides a solution. A solvent is a substance that can dissolve an insoluble solute and create a solution. The solvent is the substance that dissolves the solute and the component of a chemical solution present in the greatest amount. Solvents are often liquids, although they can also be solids, gases, or supercritical fluids. The molecules of the solvent. Solvents are often liquids, although they can also be solids, gases, or supercritical fluids. Water is a solvent for polar molecules, and the most common solvent used by living things; For example, in a saltwater mixture, water functions as the solvent, and salt is the solute. A solvent is usually a liquid, but can also be a solid or gas. A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. It is the substance in which the solute is dissolved. A solvent is a molecule that has the ability to dissolve other molecules, known as solutes. The molecules of the solvent work to put the. The solvent is the substance that dissolves the solute and the component of a chemical solution present in the greatest amount. While most common solvent are liquids, a. Usually, a solvent is a liquid. A solvent is a substance that dissolves a solute to form a homogeneous solution. A usually liquid substance capable of dissolving or dispersing one or more other substances. A solvent is the component of a solution that is present in the greatest amount. Polar solvents (e.g., water) favour formation of ion s; Something that eliminates or attenuates something.Organic_solvents Data With Water Solubility Solvent Functional Group
Solvent Solubility Table
Common Organic Solvents_ Table of Properties Solvent Functional Group
Solubility Table
Solvent Solubility Chart
Solvent Solubility Chart
Solubility Rules Chart.png
Solvent Solubility Chart
Solvent Solubility Table
Solvent Solubility Chart
A Solvent Is, By Definition, Any Substance That Will Take Other Things (Aka ‘Solutes’) Into Solution.
Solvent, Substance, Ordinarily A Liquid, In Which Other Materials Dissolve To Form A Solution.
A Solvent Can Be Solid, Liquid Or Gas.
A Solvent Is A Substance That Can Dissolve An Insoluble Solute And Create A Solution.
Related Post: