Dilution Ratio Chart
Dilution Ratio Chart - Of course, the resulting solution is thoroughly mixed so as to ensure that all parts of the solution are identical. The meaning of dilution is the action of diluting : Dilution is the addition of. Dilution is the process of weakening or deconcentrating a solution. The state of being diluted. Dilution is the process of making a concentrated solution less concentrated. The dilution ratio calculator tells you how much solute and solvent you need to get the desired dilution ratio. The process does not involve a change in the amount of solute. To dilute a solution means to add more solvent without the addition of more solute. Often, a worker will need to change the concentration of a solution by changing the amount of solvent. It is the volume of the solution that changes. Dilution is the process of decreasing the concentration of a solute in a solution, usually simply by mixing with more solvent like adding more water to the solution. Of course, the resulting solution is thoroughly mixed so as to ensure that all parts of the solution are identical. The process does not involve a change in the amount of solute. Dilution involves adding additional solvent to a sample of a solution. The state of being diluted. The dilution ratio calculator tells you how much solute and solvent you need to get the desired dilution ratio. Dilution is the process of weakening or deconcentrating a solution. For example, if you have a glass of lemonade but it is too sour, you might add more. Learn how to dilute and concentrate solutions. Often, a worker will need to change the concentration of a solution by changing the amount of solvent. For example, if you have a glass of lemonade but it is too sour, you might add more. Learn how to dilute and concentrate solutions. Dilution is the process of making a concentrated solution less concentrated. The meaning of dilution is the. Learn how to dilute and concentrate solutions. Dilution is the process of decreasing the concentration of a solute in a solution, usually simply by mixing with more solvent like adding more water to the solution. To dilute a solution means to. Dilution is the process of making a concentrated solution less concentrated. There are a variety of reasons why one. Dilution is the process of making a solution or scientific sample less concentrated by adding more solvent. Learn how to dilute and concentrate solutions. The meaning of dilution is the action of diluting : Of course, the resulting solution is thoroughly mixed so as to ensure that all parts of the solution are identical. Dilution is the process of decreasing. Dilution is the addition of. The dilution ratio calculator tells you how much solute and solvent you need to get the desired dilution ratio. For example, if you have a glass of lemonade but it is too sour, you might add more. Serial dilutions are used in microbiology to reduce bacterial concentrations to the required concentration for a particular. Dilution. The meaning of dilution is the action of diluting : To dilute a solution means to. The process does not involve a change in the amount of solute. Dilution is the process of decreasing the concentration of a solute in a solution, usually simply by mixing with more solvent like adding more water to the solution. Dilution is the process. Dilution involves adding additional solvent to a sample of a solution. Dilution is the process of weakening or deconcentrating a solution. The process does not involve a change in the amount of solute. The meaning of dilution is the action of diluting : Serial dilutions are used in microbiology to reduce bacterial concentrations to the required concentration for a particular. Dilution is the addition of. Dilution is the process of weakening or deconcentrating a solution. The meaning of dilution is the action of diluting : It is the volume of the solution that changes. Dilution is the process of decreasing the concentration of a solute in a solution, usually simply by mixing with more solvent like adding more water to. For example, if you have a glass of lemonade but it is too sour, you might add more. The process does not involve a change in the amount of solute. Serial dilutions are used in microbiology to reduce bacterial concentrations to the required concentration for a particular. Learn how to dilute and concentrate solutions. Dilution involves adding additional solvent to. The state of being diluted. Dilution is the addition of. The dilution ratio calculator tells you how much solute and solvent you need to get the desired dilution ratio. Dilution is the process of decreasing the concentration of a solute in a solution, usually simply by mixing with more solvent like adding more water to the solution. Serial dilutions are. Dilution is the addition of. There are a variety of reasons why one might want to perform a dilution. To dilute a solution means to. To dilute a solution means to add more solvent without the addition of more solute. Serial dilutions are used in microbiology to reduce bacterial concentrations to the required concentration for a particular. It is the volume of the solution that changes. Dilution is the process of weakening or deconcentrating a solution. Serial dilutions are used in microbiology to reduce bacterial concentrations to the required concentration for a particular. Dilution is the process of making a solution or scientific sample less concentrated by adding more solvent. The meaning of dilution is the action of diluting : Learn how to dilute and concentrate solutions. Apply the dilution equation to calculate the final. To dilute a solution means to. For example, if you have a glass of lemonade but it is too sour, you might add more. The state of being diluted. State whether the concentration of a solution is directly or indirectly proportional to its volume. Dilution is the process of making a concentrated solution less concentrated. Often, a worker will need to change the concentration of a solution by changing the amount of solvent. Of course, the resulting solution is thoroughly mixed so as to ensure that all parts of the solution are identical. Dilution is the process of decreasing the concentration of a solute in a solution, usually simply by mixing with more solvent like adding more water to the solution. The dilution ratio calculator tells you how much solute and solvent you need to get the desired dilution ratio.dilution ratio chart Dilution chemical ratios
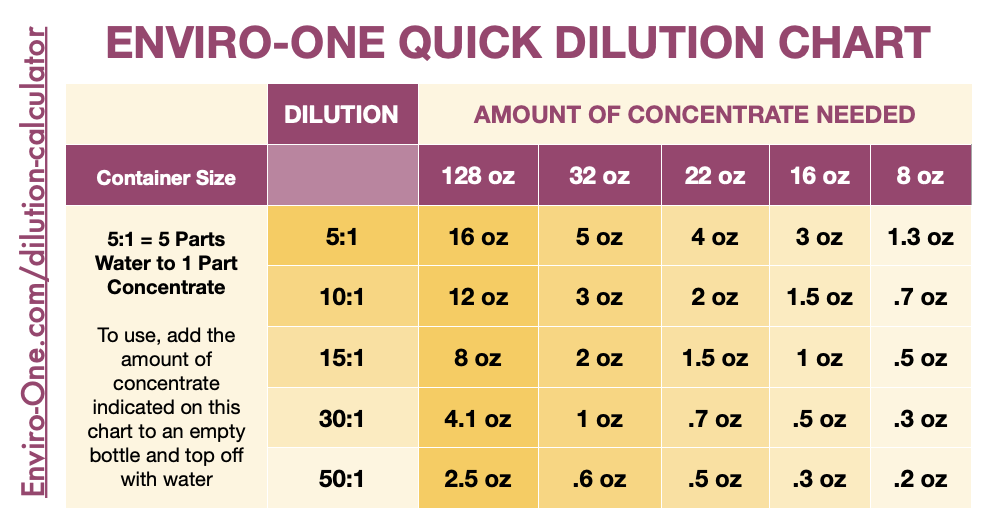
Detergent Dilution Ratios Chart Soap & Water Mix Guide
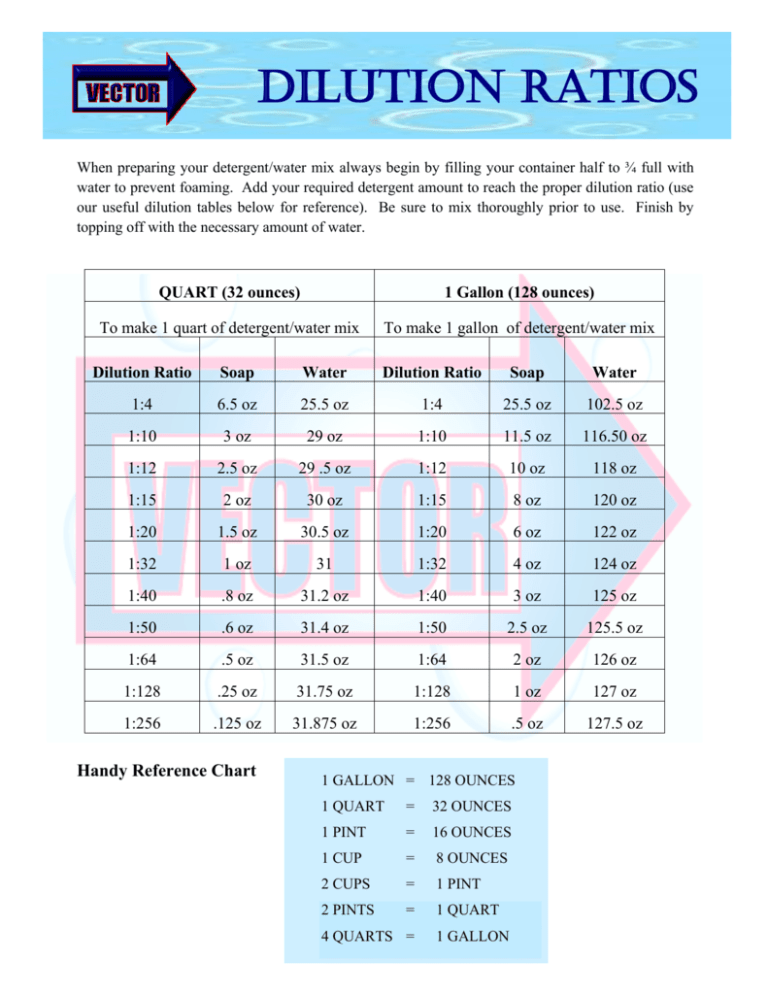
Dilution Ratio Chart A Visual Reference of Charts Chart Master
A3 Dilution Chart BICSc
Bleach Dilution Ratio Calculator at Anthony Mays blog
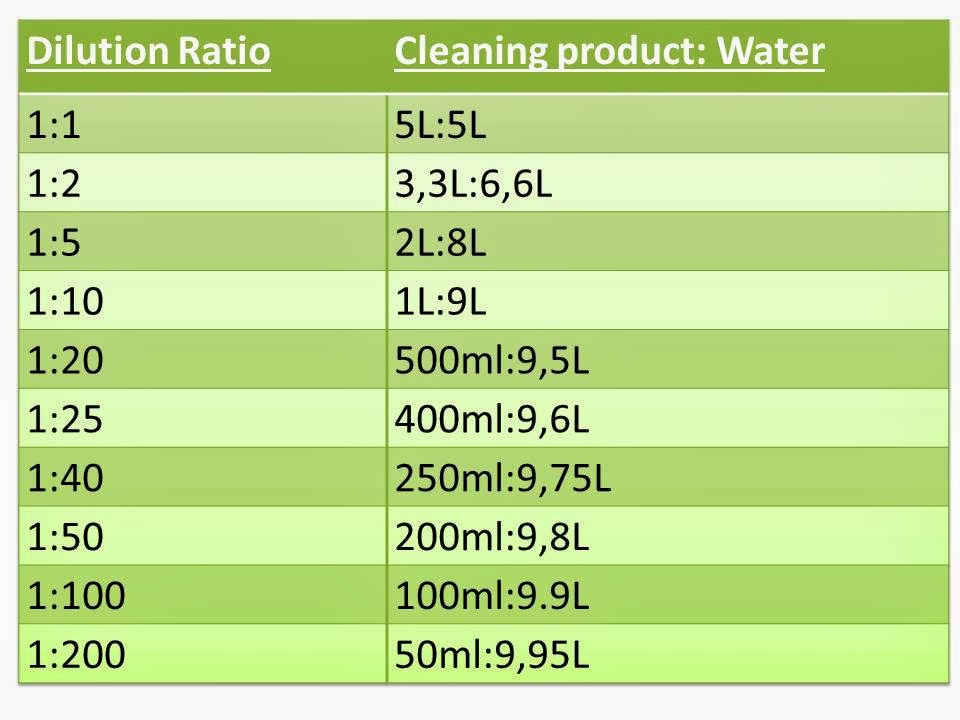
Dilution Charts Chemicals at Robert Shaver blog
CHEMICAL GUYS DILUTION CHART
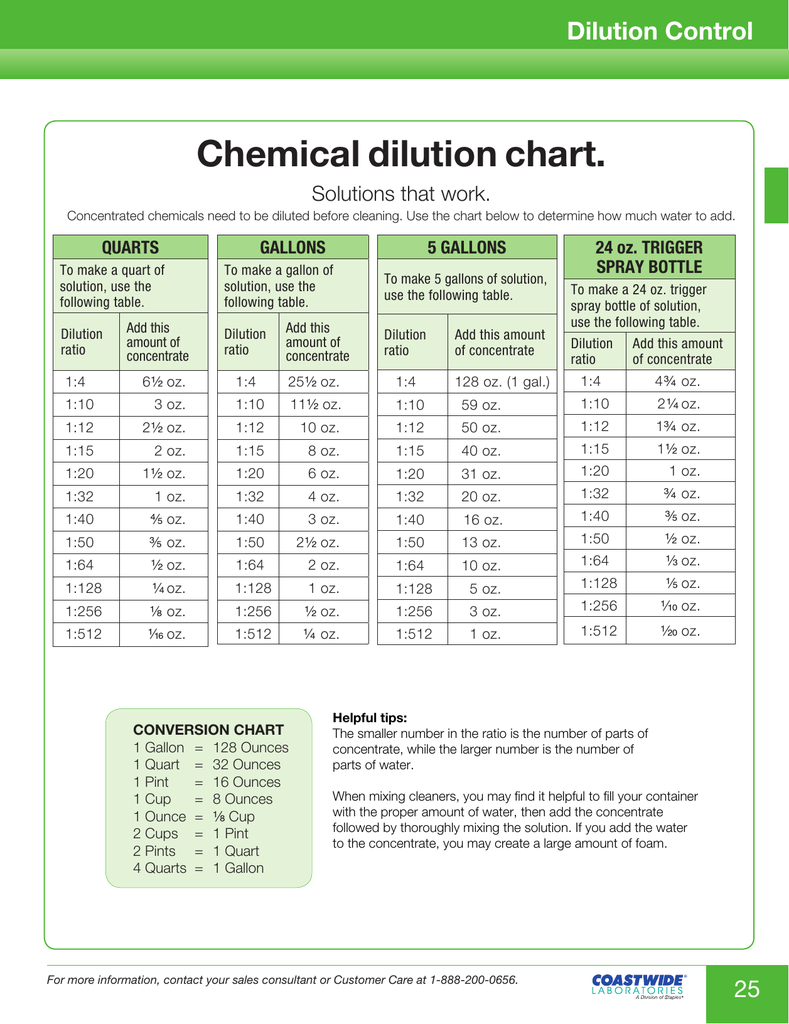
Chemical dilution chart.
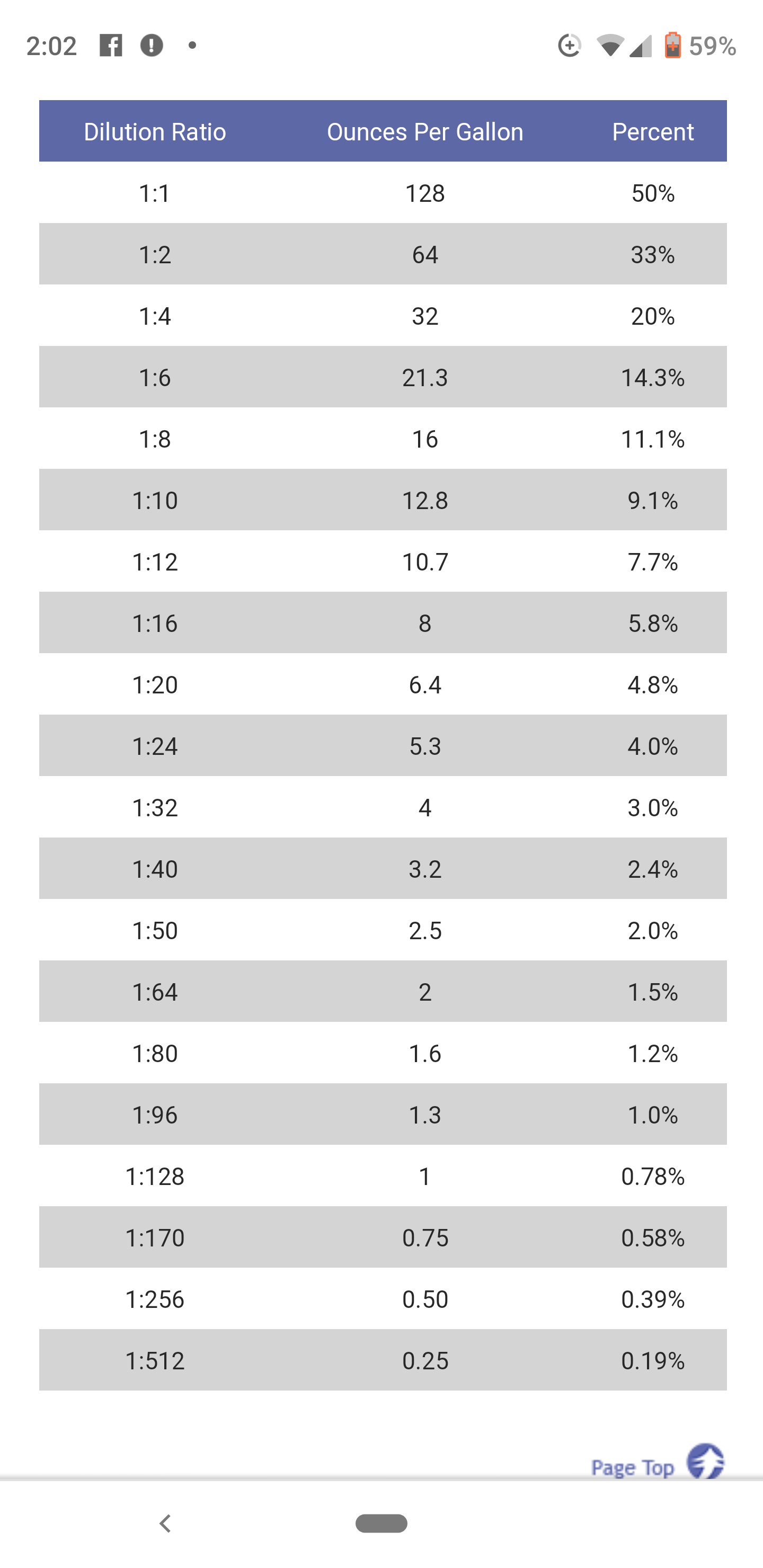
Understanding Chemical Dilution Ratios Vertech Labs
Dilution Chart.Helpful video. Understand how to prepare dilutions in Lab.Understand & clear
The Process Does Not Involve A Change In The Amount Of Solute.
To Dilute A Solution Means To Add More Solvent Without The Addition Of More Solute.
There Are A Variety Of Reasons Why One Might Want To Perform A Dilution.
Dilution Involves Adding Additional Solvent To A Sample Of A Solution.
Related Post: